-
![2-[2,6-Diisopropylphenyl]-4-phenyl-5<em>H</em>-5,9b[1′,2′]-benzonaphtho[1,2-b]pyrrol-2-ium Tetrafluoroborate](https://pub.mdpi-res.com/title_story/title_story_1690364881027.jpg?1693551999) 2-[2,6-Diisopropylphenyl]-4-phenyl-5H-5,9b[1′,2′]-benzonaphtho[1,2-b]pyrrol-2-ium Tetrafluoroborate
2-[2,6-Diisopropylphenyl]-4-phenyl-5H-5,9b[1′,2′]-benzonaphtho[1,2-b]pyrrol-2-ium Tetrafluoroborate -
 4,4′-(((Perfluoropropane-2,2-diyl)bis(4,1-phenylene))bis(oxy))-bis(2,3,5,6-tetrafluoropyridine)
4,4′-(((Perfluoropropane-2,2-diyl)bis(4,1-phenylene))bis(oxy))-bis(2,3,5,6-tetrafluoropyridine) -
 Synthesis, Characterization and Chemistry of Tetrakis(Propargylisocyanide) Copper(I) Complex
Synthesis, Characterization and Chemistry of Tetrakis(Propargylisocyanide) Copper(I) Complex -
![2-(3-Bromophenyl)imidazo[2,1-<em>b</em>]oxazole](https://pub.mdpi-res.com/title_story/title_story_16892324932128.jpg?1693551999) 2-(3-Bromophenyl)imidazo[2,1-b]oxazole
2-(3-Bromophenyl)imidazo[2,1-b]oxazole -
 3-Cyclohexyl-6-phenyl-1-(p-tolyl)pyrimidine-2,4(1H,3H)-dione
3-Cyclohexyl-6-phenyl-1-(p-tolyl)pyrimidine-2,4(1H,3H)-dione
Journal Description
Molbank
Molbank
is a peer-reviewed, open access journal comprised of a unique collection of one-compound-per-paper short notes on synthetic compounds and natural products. Molbank has been published online quarterly since 2009 (previously published irregularly).
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), Reaxys, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.9 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the first half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
0.6 (2022)
Latest Articles
(2R,4aS,6aS,12bR,14aS,14bR)10-Hydroxy-N-(4-((6-methoxyquinolin-8-yl)amino)pentyl)-2,4a,6a,9,12b,14a-hexamethyl-11-oxo-1,2,3,4,4a,5,6,6a,11,12b,13,14,14a,14b-tetradecahydropicene-2-carboxamide
Molbank 2023, 2023(3), M1716; https://doi.org/10.3390/M1716 - 29 Aug 2023
Abstract
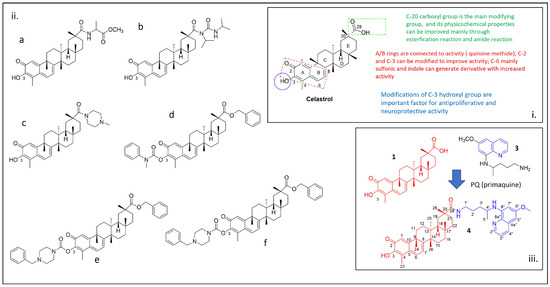
We herein report the synthesis of a derivative of the natural compound celastrol linked to the antimalarial drug primaquine through an amide obtained by the activation of the carboxylic acid with HOBt/EDC. The chemical structure of the new molecule was fully characterized by
[...] Read more.
We herein report the synthesis of a derivative of the natural compound celastrol linked to the antimalarial drug primaquine through an amide obtained by the activation of the carboxylic acid with HOBt/EDC. The chemical structure of the new molecule was fully characterized by proton nuclear magnetic resonance (1H-NMR), carbon-13 nuclear magnetic resonance (13C-NMR), heteronuclear single quantum coherence (HSQC), correlation spectroscopy (1H-1H-COSY), distortionless enhancement by polarization transfer (DEPT), mass spectrometry, Fourier-transform infrared (FTIR), and ultraviolet (UV) spectroscopies. Computational studies were enrolled to predict the interaction of the synthesized compound with sarco-endoplasmic reticulum (SR) Ca2+ transport ATPase (SERCA), a target of relevance for developing new therapeutics against arthritis. The drug-likeness of the compound was also investigated by predicting its pharmacokinetic properties.
Full article
(This article belongs to the Section Organic Synthesis)
►
Show Figures
Open AccessCommunication
Synthesis of a Novel 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde as a Bisulfite Adduct
Molbank 2023, 2023(3), M1715; https://doi.org/10.3390/M1715 - 24 Aug 2023
Abstract
►▼
Show Figures
The scope of the current work was to synthesize an S-alkylated 1,2,4-triazole-3-thiol derivative. Synthesis was carried out in two steps: in the first step, 4,5-diphenyl-4H-1,2,4-triazole-3-thiol was S-alkylated using a halogenated acetal and cesium carbonate. In the second step, several
[...] Read more.
The scope of the current work was to synthesize an S-alkylated 1,2,4-triazole-3-thiol derivative. Synthesis was carried out in two steps: in the first step, 4,5-diphenyl-4H-1,2,4-triazole-3-thiol was S-alkylated using a halogenated acetal and cesium carbonate. In the second step, several acetal deprotection procedures were tested, and the aldehyde obtained was isolated as a bisulfite adduct. The structures of the new compounds were characterized by FT-IR, 1D, and 2D NMR spectroscopic methods.
Full article

Graphical abstract
Open AccessShort Note
(E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile
by
, , , , , and
Molbank 2023, 2023(3), M1714; https://doi.org/10.3390/M1714 - 21 Aug 2023
Abstract
►▼
Show Figures
[1,2,5]Oxadiazolo[3,4-b]pyrazines are of great interest due to their promising photophysical and electrochemical properties, as well their potential use in a wide range of electronic devices. Herein, we report on the preparation of the unexpected product derived from the interaction of 2′-([1,2,5]oxadiazolo[3,4-
[...] Read more.
[1,2,5]Oxadiazolo[3,4-b]pyrazines are of great interest due to their promising photophysical and electrochemical properties, as well their potential use in a wide range of electronic devices. Herein, we report on the preparation of the unexpected product derived from the interaction of 2′-([1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)-N,N-diphenyl-[1,1′-biphenyl]-4-amine with 2-cyanoacetic acid under basic conditions. The resulting product was characterized using 1H and 13C NMR spectra, high resolution mass spectrometry, Fourier-transform infrared spectroscopy (FTIR), and X-ray diffraction analyses. Furthermore, its photophysical and electrochemical properties were studied using cyclic voltammetry, UV–Vis, and emission spectroscopy. The experimental results have been further rationalized through theoretical DFT calculations.
Full article

Figure 1
Open AccessCommunication
Friedländer-Type Reaction of 4-Cholesten-3-one with 2′-Aminoacetophenone: Angular versus Linear Quinoline-Fused Steroids
Molbank 2023, 2023(3), M1712; https://doi.org/10.3390/M1712 - 16 Aug 2023
Abstract
To optimize the experimental conditions used for the Friedländer-type condensation, an angular fused 4-substituted quinoline steroid has been obtained in very high yield and regioselectivity using readily available 4-cholesten-3-one and 2′-aminoacetophenone. Moreover, by varying the reaction conditions and the catalyst, the corresponding linear
[...] Read more.
To optimize the experimental conditions used for the Friedländer-type condensation, an angular fused 4-substituted quinoline steroid has been obtained in very high yield and regioselectivity using readily available 4-cholesten-3-one and 2′-aminoacetophenone. Moreover, by varying the reaction conditions and the catalyst, the corresponding linear regioisomer was also achieved with an acceptable isolated yield and high chemoselectivity. Both structures have been definitively elucidated via 2D-NMR and fully characterized.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures
Figure 1
Open AccessCommunication
Synthesis of a New α-Azidomethyl Styrene from Safrole via a Dearomative Rearrangement
Molbank 2023, 2023(3), M1713; https://doi.org/10.3390/M1713 - 16 Aug 2023
Abstract
There is a growing interest in developing more efficient synthetic alternatives for the synthesis of nitrogen-containing allylic compounds. This article presents a straightforward two-step protocol to produce 5-(3-azidoprop-1-en-2-yl)benzo[d][1,3]dioxole 4 from the natural product safrole. The method yielded the expected α-azidomethyl styrene
[...] Read more.
There is a growing interest in developing more efficient synthetic alternatives for the synthesis of nitrogen-containing allylic compounds. This article presents a straightforward two-step protocol to produce 5-(3-azidoprop-1-en-2-yl)benzo[d][1,3]dioxole 4 from the natural product safrole. The method yielded the expected α-azidomethyl styrene 4, in good yield, via a dearomative rearrangement.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Scheme 1
Open AccessShort Note
Pentapotassium Bis(hydrogenphospate) Dihydrogenphospate Monohydrate
Molbank 2023, 2023(3), M1711; https://doi.org/10.3390/M1711 - 14 Aug 2023
Abstract
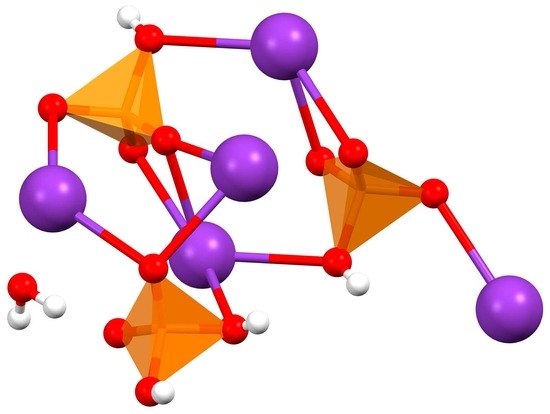
The structure of K5(HPO4)2(H2PO4)·H2O was determined via single crystal diffraction. The crystal structures of phosphate salts of potassium have been known since the early days of crystallography. Here, we present a new monohydrate adduct between K2HPO4 and KH2PO4.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
Graphical abstract
Open AccessShort Note
Tributyl(1-((dimethylamino)(dimethyliminio)methyl)-1,4-dihydropyridin-4-yl)phosphonium Ditrifluoromethanesulfonate
Molbank 2023, 2023(3), M1710; https://doi.org/10.3390/M1710 - 09 Aug 2023
Abstract
►▼
Show Figures
Site-selective functionalization of pyridines is a crucial tool for the synthesis of diverse pharmaceuticals and materials. We introduced diiminium pyridine adducts as highly convenient and potent Lewis acids. We report that tributylphosphine selectively adds to the 4-position of pyridine in tetramethyldiiminium pyridine ditrifluoromethanesulfonate,
[...] Read more.
Site-selective functionalization of pyridines is a crucial tool for the synthesis of diverse pharmaceuticals and materials. We introduced diiminium pyridine adducts as highly convenient and potent Lewis acids. We report that tributylphosphine selectively adds to the 4-position of pyridine in tetramethyldiiminium pyridine ditrifluoromethanesulfonate, resulting in the formation of the title compound. This finding represents an advancement towards the utilization of diiminium units as organic reagents or catalysts for pyridine functionalization. We also employ computational models to determine fluoride and hydride ion affinities, Fukui function f+(r), molecular electrostatic potential, and pKa values, providing valuable insights for future investigations in this area.
Full article
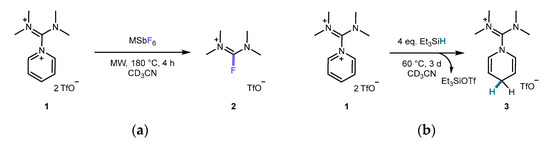
Figure 1
Open AccessShort Note
(E)-2-(1,3-Diphenylallyl)-3,5-dimethoxyphenol
Molbank 2023, 2023(3), M1709; https://doi.org/10.3390/M1709 - 04 Aug 2023
Abstract
►▼
Show Figures
The synthesis of (E)-2-(1,3-diphenylallyl)-3,5-dimethoxyphenol is described by means of the reaction of 3,5-dimethoxyphenol with (E)-1,3-diphenylprop-2-en-1-ol in 1,1,1,3,3,3-hexafluoroispropanol (HFIP), which acts as a solvent and reaction promoter. The reaction proceeds smoothly to afford the mentioned compound in high yield under
[...] Read more.
The synthesis of (E)-2-(1,3-diphenylallyl)-3,5-dimethoxyphenol is described by means of the reaction of 3,5-dimethoxyphenol with (E)-1,3-diphenylprop-2-en-1-ol in 1,1,1,3,3,3-hexafluoroispropanol (HFIP), which acts as a solvent and reaction promoter. The reaction proceeds smoothly to afford the mentioned compound in high yield under a metal and additive-free procedure. The corresponding allylated phenol has been fully characterized.
Full article

Figure 1
Open AccessShort Note
Isoamyl (E)-3-(3,4-Dimethoxyphenyl)acrylate
Molbank 2023, 2023(3), M1708; https://doi.org/10.3390/M1708 - 02 Aug 2023
Abstract
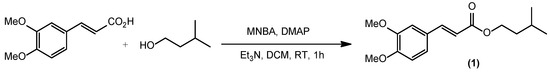
Isoamyl (E)-3-(3,4-dimethoxyphenyl)acrylate (1) was obtained from the 2-methyl-6-nitrobenzoic anhydride (MNBA)/4-dimethylaminopyridine (DMAP)-catalyzed reaction at room temperature for 190 min in dichloromethane with a yield of 95%. The structure of isoamyl (E)-3-(3,4-dimethoxyphenyl)acrylate (1) was elucidated using NMR,
[...] Read more.
Isoamyl (E)-3-(3,4-dimethoxyphenyl)acrylate (1) was obtained from the 2-methyl-6-nitrobenzoic anhydride (MNBA)/4-dimethylaminopyridine (DMAP)-catalyzed reaction at room temperature for 190 min in dichloromethane with a yield of 95%. The structure of isoamyl (E)-3-(3,4-dimethoxyphenyl)acrylate (1) was elucidated using NMR, FTIR, and high-resolution mass spectrometry. In vitro sun protection factor evaluation exhibited a value of 37.10 ± 0.03 which indicates that isoamyl (E)-3-(3,4-dimethoxyphenyl)acrylate (1) is a sunscreen agent with high protection.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures
Scheme 1
Open AccessShort Note
N′-(5-Bromofuran-2-carbonyl)isonicotinohydrazide
Molbank 2023, 2023(3), M1706; https://doi.org/10.3390/M1706 - 01 Aug 2023
Abstract
N′-(5-bromofuran-2-carbonyl)isonicotinohydrazide (1) was obtained in the form of a colorless solid from the 2-methyl-6-nitrobenzoic anhydride (MNBA)/4-dimethylaminopyridine (DMAP)-catalyzed reaction of 5-bromofuran-2-carboxylic acid and isoniazid in dichloromethane at room temperature with a yield of 83%. The structure of N′-(5-bromofuran-2-carbonyl)isonicotinohydrazide (1
[...] Read more.
N′-(5-bromofuran-2-carbonyl)isonicotinohydrazide (1) was obtained in the form of a colorless solid from the 2-methyl-6-nitrobenzoic anhydride (MNBA)/4-dimethylaminopyridine (DMAP)-catalyzed reaction of 5-bromofuran-2-carboxylic acid and isoniazid in dichloromethane at room temperature with a yield of 83%. The structure of N′-(5-bromofuran-2-carbonyl)isonicotinohydrazide (1) was elucidated using 1H NMR, 13C NMR, FTIR, and high-resolution mass spectrometry. Molecular docking screening of the title compound (1) on cyclooxygenase-2 (COX-2) protein (PDB ID: 5IKR) indicated that compound (1) has a good binding affinity, suggesting that further structure optimization and in-depth research can be carried out on compound (1) as a potential COX-2 inhibitor.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Figure 1
Open AccessCommunication
Synthesis of Novel Diterpenic Peptides via the Ugi Reaction and Their Anticancer Activities
Molbank 2023, 2023(3), M1707; https://doi.org/10.3390/M1707 - 01 Aug 2023
Abstract
Novel diterpenic peptide derivatives were synthesized via the Ugi four-component reaction at ambient temperature. The protocol employed a reaction between formaldehyde, benzyl amine, the corresponding diterpene acid, and ethyl 2-isocyanoacetate. The anticancer properties of the compounds were studied in vitro.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Scheme 1
Open AccessShort Note
4-(4-ethoxyphenyl)-5-(4-methoxyphenyl)-2,4-dihydro-3H-1,2,4-triazol-3-one
Molbank 2023, 2023(3), M1705; https://doi.org/10.3390/M1705 - 01 Aug 2023
Abstract
A new triazol-3-one resulted unexpectedly from the reduction reaction of a heterocyclic thioketone using sodium borohydride in pyridine containing a small amount of water. The structure of the new compound was characterised using FT-IR, 1D and 2D NMR, and HRMS spectroscopic methods.
Full article
(This article belongs to the Special Issue Molecules from Side Reactions II)
►▼
Show Figures

Graphical abstract
Open AccessCommunication
N-(((1S,5R)-6,6-Dimethylbicyclo[3.1.1]hept-2-en-2-yl)methyl)-3-dodecan/tetradecanamido-N,N-dimethylpropan-1-aminium Bromide
by
, , , , , , , , , , , and
Molbank 2023, 2023(3), M1704; https://doi.org/10.3390/M1704 - 26 Jul 2023
Abstract
The syntheses of the title compounds were performed using lauric and myristic acids. The compounds obtained were characterized using 1H-, 13C-NMR and 2D 1H-1H COSY, 1H-13C HSQC NMR, IR, and high-resolution mass spectrometry. Both compounds
[...] Read more.
The syntheses of the title compounds were performed using lauric and myristic acids. The compounds obtained were characterized using 1H-, 13C-NMR and 2D 1H-1H COSY, 1H-13C HSQC NMR, IR, and high-resolution mass spectrometry. Both compounds exhibited bactericidal activity on S. aureus comparable to that of a reference drug (miramistin). Compound 10, with lauric acid fragment, had a 16-fold higher activity on P. aeruginosa compared to compound 11, which in turn contains myristic acid fragment (with minimum inhibitory concentrations of 32 and 512 μg/mL, respectively). Compound 11 exhibited a pronounced activity against all types of fungi (higher than the activity of miramistin), while the activity of compound 10 was considerably lower. Thus, compound 11 can serve as a promising antimicrobial agent for the treatment of various fungal and staphylococcal infections, while compound 10 is of interest to treat P. aeruginosa-associated infections.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Graphical abstract
attachment
Supplementary material:
Supplementary File 1 (ZIP, 622 KiB)
Supplementary File 2 (MOL, 5 KiB)
Supplementary File 3 (INCHI, 3 KiB)
Supplementary File 4 (MOL, 5 KiB)
Supplementary File 5 (MOL, 3 KiB)
Supplementary File 6 (INCHI, 2 KiB)
Supplementary File 7 (MOL, 4 KiB)
Supplementary File 8 (CIF, 1130 KiB)
Supplementary File 1 (ZIP, 622 KiB)
Supplementary File 2 (MOL, 5 KiB)
Supplementary File 3 (INCHI, 3 KiB)
Supplementary File 4 (MOL, 5 KiB)
Supplementary File 5 (MOL, 3 KiB)
Supplementary File 6 (INCHI, 2 KiB)
Supplementary File 7 (MOL, 4 KiB)
Supplementary File 8 (CIF, 1130 KiB)
Open AccessShort Note
2,7-Bis(pyridin-4-ylethynyl)-9H-carbazole
by
, , , , , , and
Molbank 2023, 2023(3), M1703; https://doi.org/10.3390/M1703 - 24 Jul 2023
Abstract
2,7-Bis(pyridin-4-ylethynyl)-9H-carbazole (1) was synthesized by reacting 4-ethynylpyridine hydrochloride with 2,7-dibromo-9H-carbazole. The full characterization of compound 1 is presented, and the crystal structure of its monohydrate was determined by single-crystal XRD analysis.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
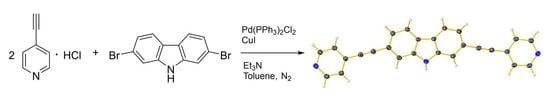
Graphical abstract
Open AccessShort Note
6,7-Dihydroxy-5,8-dimethoxy-2H-chromen-2-one
Molbank 2023, 2023(3), M1702; https://doi.org/10.3390/M1702 - 23 Jul 2023
Abstract
This article presents a novel approach for synthesizing a new 5,8-dimethoxy derivative of esculetin via the selective cleavage of the methylene bridge in sabandin—naturally occurring and easily synthetically accessible methoxylated coumarin. A high selectivity is achieved by using acetoxylation of methylenedioxy group with
[...] Read more.
This article presents a novel approach for synthesizing a new 5,8-dimethoxy derivative of esculetin via the selective cleavage of the methylene bridge in sabandin—naturally occurring and easily synthetically accessible methoxylated coumarin. A high selectivity is achieved by using acetoxylation of methylenedioxy group with lead tetraacetate. Natural coumarin sabandin as a starting compound was prepared in a few simple steps from 5-allyl-4,7-dimethoxybenzo[d][1,3]dioxole (apiol), which is readily available from parsley and dill seed extracts. The developed method enables an efficient and straightforward synthesis of a new derivative of esculetin with potential medicinal and therapeutic applications.
Full article
(This article belongs to the Section Organic Synthesis)
►▼
Show Figures

Graphical abstract
Open AccessShort Note
5-((4-(-Phenyldiazenyl)phenyl)diazenyl)quinolin-8-ol
Molbank 2023, 2023(3), M1701; https://doi.org/10.3390/M1701 - 21 Jul 2023
Abstract
►▼
Show Figures
A new azo compound was synthesized via an azo coupling reaction between 4-(phenyldiazenyl)benzenediazonium chloride and 8-hydroxyquinoline (8-Hq). The new diazene compound can be used to synthesize metal complexes as a derivative of 8-Hq. The structure of the new compound was characterized using UV–Vis,
[...] Read more.
A new azo compound was synthesized via an azo coupling reaction between 4-(phenyldiazenyl)benzenediazonium chloride and 8-hydroxyquinoline (8-Hq). The new diazene compound can be used to synthesize metal complexes as a derivative of 8-Hq. The structure of the new compound was characterized using UV–Vis, FT-IR, and 2D NMR spectroscopic methods.
Full article

Scheme 1
Open AccessShort Note
(2S,2’S,4R,5S,5’R)-2,2’-Di-tert-butyl-4-hydroxy-5,5’-dimethyl-4,5’-bi(1,3-dioxolanyl)-4’-one
Molbank 2023, 2023(3), M1699; https://doi.org/10.3390/M1699 - 20 Jul 2023
Abstract
The product formed by base-induced dimerisation of (2S,5S)-2-tert-butyl-5-methyl-1,3-dioxolan-4-one is shown by X-ray diffraction to be the title compound and not the isomeric fused-ring 1,3-dioxolane/1,3-dioxane-4-one structure proposed by previous researchers. The analogous compound derived from (2S,5S)-5-benzyl-2-tert-butyl-1,3-dioxolan-4-one has also been obtained and characterised.
Full article
(This article belongs to the Section Structure Determination)
►▼
Show Figures
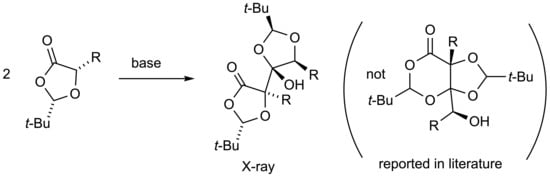
Graphical abstract
Open AccessShort Note
4-(2,5-Dimethyl-1H-pyrrol-1-yl)-1,2,5-oxadiazol-3-amine
Molbank 2023, 2023(3), M1700; https://doi.org/10.3390/M1700 - 20 Jul 2023
Abstract
1,2,5-Oxadiazol-3-amines with a heterocyclic substituent in the 4-position are being intensively investigated as compounds with valuable pharmacological activity. In this communication, the reaction of 1,2,5-oxadiazole-3,4-diamine with 2,5-hexanedione was shown to selectively give 4-(2,5-dimethyl-1H-pyrrol-1-yl)-1,2,5-oxadiazol-3-amine as a product of the Paal–Knorr reaction. The
[...] Read more.
1,2,5-Oxadiazol-3-amines with a heterocyclic substituent in the 4-position are being intensively investigated as compounds with valuable pharmacological activity. In this communication, the reaction of 1,2,5-oxadiazole-3,4-diamine with 2,5-hexanedione was shown to selectively give 4-(2,5-dimethyl-1H-pyrrol-1-yl)-1,2,5-oxadiazol-3-amine as a product of the Paal–Knorr reaction. The structure of the synthesized compound was established by elemental analysis, high-resolution mass spectrometry, 1H and 13C NMR, and IR spectroscopy.
Full article
(This article belongs to the Collection Heterocycle Reactions)
►▼
Show Figures
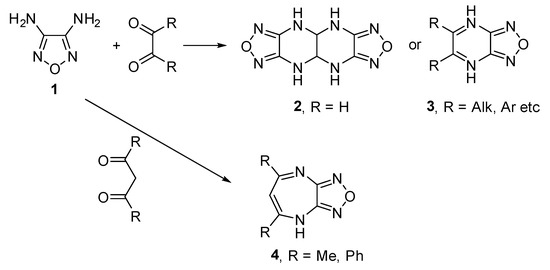
Scheme 1
Open AccessShort Note
Methyl-4-hydroxy-2-(2-hydroxypropan-2-yl)-6-methyl-2,3-dihydrobenzofuran-5-carboxylate
Molbank 2023, 2023(3), M1697; https://doi.org/10.3390/M1697 - 19 Jul 2023
Abstract
Microorganisms are an important source of compounds that are pharmaceutically active, both as leads and as scaffolds for synthesis. Often, interesting chemistry is uncovered by exploring new biomes, of which the Chilean Atacama Desert is a prime example. This paper describes the isolation
[...] Read more.
Microorganisms are an important source of compounds that are pharmaceutically active, both as leads and as scaffolds for synthesis. Often, interesting chemistry is uncovered by exploring new biomes, of which the Chilean Atacama Desert is a prime example. This paper describes the isolation and structural characterisation, using HR-LCMS and 1D and 2D NMR, of a new compound belonging to a family of compounds called radstrictins. The compound was isolated from a fungus, that had itself been isolated from a soil sample from the Atacama Desert. The compound was tested against pathogenic strains associated with bovine mastitis, but was found to be devoid of antimicrobial activity.
Full article
(This article belongs to the Section Natural Products)
►▼
Show Figures

Figure 1
Open AccessShort Note
[μ-1,2-Bis(dipheylphosphino)ethane-κ2P,P’]bis(3-mercapto-1,2-propanediolato-κS-gold(I))
by
and
Molbank 2023, 2023(3), M1698; https://doi.org/10.3390/M1698 - 19 Jul 2023
Abstract
►▼
Show Figures
A new dinuclear gold(I) complex, possessing a bridging diphosphine ligand (1,2-bis(diphenylphosphino)ethane) and two terminal thiol ligands (1-thioglycerol), was synthesized and fully characterized by IR, 1H and 31P NMR, fluorescence, ESI-mass, and diffuse reflection spectroscopy, together with X-ray diffraction and elemental analyses.
[...] Read more.
A new dinuclear gold(I) complex, possessing a bridging diphosphine ligand (1,2-bis(diphenylphosphino)ethane) and two terminal thiol ligands (1-thioglycerol), was synthesized and fully characterized by IR, 1H and 31P NMR, fluorescence, ESI-mass, and diffuse reflection spectroscopy, together with X-ray diffraction and elemental analyses. The compound formed a 1D chain supramolecular structure through intermolecular aurophilic interactions in the crystal structure, leading to photoluminescence in the solid state.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Catalysts, Chemistry, Molbank, Molecules, Sustainable Chemistry
Towards the Sustainable Synthesis of Biologically Active Molecules in Green Solvents
Topic Editors: Antonio Salomone, Serena PerroneDeadline: 31 December 2024